KoFaLiS
Development of innovative Li2S composite materials to increase cycle stability and high current capability of lithium-sulfur batteries
Technological advances, especially in portable and handheld devices are unthinkable without the development in the battery technologies during the past few decades. There are many battery technologies out there, although Lithium-ion (Li-ion) batteries are the ones mostly and widely used now a days. However, Li-ion battery technology is reaching its practical and theoretical specific energy limits of 200-250 Wh/kg under new applied situations such as electrical vehicles and unmanned aerial vehicles where light weight batteries with high energy densities are needed.
Among the rechargeable batteries, Lithium-sulfur (LiS) batteries have a potential to completely replace Li-ion batteries. They outperform Li-ion batteries in terms of energy densities and reduced cost, due to use of abundant sulfur. The fact that heavy, expensive and sometimes toxic metal-oxides’ cathode is replaced by low-atomic weight sulfur, LiS batteries are also light weight at the same time. The theoretical energy density of LiS batteries is up to five times higher than the commercial Li-ion batteries. Still, LiS battery commercialization is hindered by challenges within the cell. Poor electrical conductivity of initial S8 cathode along with the use of heavy weight Li-foil as anode are among them. Therefore, either conductive additives and binders are added to cathode or coatings are made to improve the electrical conductivity of cathode. That leads to another problem, which is 80% volume expansion of sulfur cathode when S8 changes to Li2S during Li-ion loading and vice versa. Such mechanical stress leads to the destruction of electrode or delamination of the coating from current collector and thus results in decreased capacity and even the failure of the battery cell. Current studies for LSB with a Li2S cathode indicate a high achievable capacitance at low current load, but the capacitance drops significantly at a medium as well as high current load. Long-term stability also needs to be significantly improved before LSB is ready for the market.
Furthermore, electrolyte soluble lithium polysulfides are formed during the cell operation. These dissolved polysulfides lead to undesirable side reactions in the battery cell, resulting in a decrease capacity, low Coulomb efficiency, increase in resistance and short battery life.
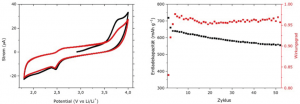
Link: Cyclic voltammograms of the electrode of carbon nanofibers with a scan rate of 50 µV/s in the voltage range from 1.85 V to 4.0 V. Right: galvanostatic cycling performance of the electrode of carbon nanofibers from the 1st to the 50 cycles performed in a CR2032 coin cell with a glass fiber separator and in a voltage range from 1.85 V to 2.6 V at a current rate of 0.1 C.
In this project, customized current collectors based on carbon nanofibers (CNF) will be developed for LSB by electrospinning technique at DTNW. Compared to metal foils as current collectors, a pore-rich, three-dimensional conductive structure of the current collector is intended to effectively compensate for the mechanical stresses occurring during cell operation.
By including the right additives in spinning dope and choosing the optimum spinning parameters, the surface area and the pore size of the CNFs will be optimized for the application. By introducing precursors for transition metal oxides (e.g. TiO2 or MnO2), sulfides (e.g. MoS2 or WS2) and metallic nanoparticles (e.g. Ag) during the spinning process, both the electrochemical and electrical conductivity of the electrode will be further improved and any polysulfides formed can be effectively absorbed.
LSB cells will be assembled and electrochemically analyzed at ZBT. Initially, the pore geometry of the current collector will be used to selectively adjust the particle size of Li2S. In the further course of the project, Li2S will be modified with carbon and transition metal sulfides to improve the high-current capability as well as the stability of Li2S cathodes. Subsequently, electrodes will be fabricated and electrochemically investigated. These investigations will be combined with in situ UV-VIS measurements to verify the effectiveness of the planned polysulfide adsorption measures.
The goals of the project are to achieve the following
- An initial cell capacity of at least 900 mAh g-1 (measured at a low current load of 0.05 C)
- At least 85% of the initial cell capacity retention after 500 cycles
- An average Coulomb efficiency of more than 98.5%
- At least 75% of the initial capacitance at 1 C (medium current load) and 50% of the initial capacitance at 5 C (high current load
Project Information:
Title (German): Entwicklung innovativer Li2S-Kompositmaterialien zur Steigerung der Zyklenfestigkeit und Hochstromfähigkeit von Lithium-Schwefel-Batterien
Acronym: KoFaLiS
Grant: 21731 N
Runtime: 01.03.2021 bis 31.08.2023
Project Partners: Deutsches Textilforschungszentrum Nord-West gGmbH, Krefeld
Hochschule Reutlingen, Reutlingen Research Institute/ Lehr- und Zentrum für Brennstoffzellentechnik GmbH (ZBT), Duisburg
Contact DTNW: Dr. Andreas Wego, Tel.: +49-2151-843-2017, E-Mail: wego@dtnw.de
Weitere Informationen zum Download:




